|
NORMAL ACID-BASE REGULATION
An ion is a substance that can carry an electrical charge. The acid-base balance is defined by the concentration of hydrogen ions (the pH). The normal pH of the body is very slightly alkaline. Acidic substances in the body include carbon dioxide and molecules containing hydrogen ions. These are buffered (counteracted) by alkaline substances, primarily bicarbonate, with some buffering by phosphate, proteins, hemoglobin, and other substances. Increased carbon dioxide or decreased bicarbonate levels can create an acidic state in the body, called acidosis. Decreased carbon dioxide or increased bicarbonate levels can create an excess alkaline state, called alkalosis. If the defect is caused by changes in carbon dioxide levels, it is a respiratory acidosis or respiratory alkalosis. If the defect is caused by changes in bicarbonate levels, it is a metabolic acidosis or metabolic alkalosis.
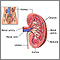
The respiratory system regulates the levels of carbon dioxide through changes in the breathing rate. Carbon dioxide is lost with faster breathing and increased when breathing slows. This provides fast, but temporary regulation of body pH.
The major, long-term regulation of body pH occurs in the kidneys, which excrete acids, and excrete or create bicarbonate for use in the body.
DISTAL RENAL TUBULAR ACIDOSIS
Renal tubular acidosis is one common cause of metabolic acidosis. Distal renal tubular acidosis (Type I RTA) is a disorder caused by a defect in the secretion of hydrogen ions in the distalrenal tubule (the late portion of the kidney tubule). This causes a reduction in the reabsorption of bicarbonate into the bloodstream.
Type I RTA is caused by a variety of conditions including hereditary disorders, autoimmune diseases and certain drugs including amphotericin B, lithium, and analgesics.
Renal tubular acidosis causes disorders of the body related to the loss of bicarbonate and inability to excrete hydrogen. The body attempts to maintain an electro-chemical balance between positively charged and negatively charged molecules. If the excretion of a molecule is abnormal, the excretion of other molecules may become abnormal in an attempt to maintain a balance.
Type I RTA causes retention of acid and is also associated with mild loss of potassium in the urine.
The acidic condition of the body causes calcium to dissolve from the bones. The calcium accumulates in the bloodstream, and excess serum calcium is excreted by the kidneys, causing a loss of total body calcium and resulting in osteomalacia or rickets, impaired growth of children, skeletal deformities, and muscle weakness. There is an increased incidence of kidney stones and nephrocalcinosis associated with the excessive excretion of calcium and phosphate through the kidneys.
|